Dr Martin Juneau, M.D., FRCP
Cardiologue, directeur de l'Observatoire de la prévention de l'Institut de Cardiologie de Montréal. Professeur titulaire de clinique, Faculté de médecine de l'Université de Montréal. / Cardiologist and Director of Prevention Watch, Montreal Heart Institute. Clinical Professor, Faculty of Medicine, University of Montreal.
See all articlesOverview
- Although the recommended dietary protein intake is 0.8-1.0 g per kg per day, which corresponds to approximately 10-15% of total calories, protein intake is much higher in some people and can even exceed 20% of calories.
- The impact of this high protein intake was examined by comparing the effect of a meal containing normal (15% of total calories) or high amounts of protein (22% of calories) on macrophage function, a class of immune cells involved in the development of atherosclerosis.
- The results indicate that the protein-rich meal causes an overactivation of the macrophage mTOR complex, which is correlated with an acceleration of the formation of atherosclerotic plaques.
- The amino acid leucine, which is mainly present in animal proteins, can reproduce the atherogenic effect of large quantities of protein.
- A very high intake of animal protein, about twice the recommended intake, could therefore promote the development of atherosclerosis and increase the risk of cardiovascular disease.
Protein is one of the most important components of our diet because it contains essential amino acids that we are unable to produce ourselves, and that absolutely must be provided by what we eat. These amino acids are used by our metabolism to manufacture tens of thousands of different proteins, which all contribute in their own way to optimal human functioning, whether as enzymes, hormones, neurotransmitters, structural proteins, receptors, or detectors of pathogens (among others). Consequently, while we have witnessed the emergence over the years of diets that drastically reduce carbohydrate intake (the ketogenic diet, for example) or fat intake (low-fat vegan diets), there are no (and never will be) diets based on reducing protein intake below recommended minimums. On the contrary, studies show that most people perceive protein-rich foods or those with added protein as “health foods”, and they try to “eat more protein”.
Recommended dietary allowance
The amount of protein required to meet the human body’s needs is not that difficult to obtain. Studies have shown that the minimum protein intake necessary to offset losses associated with normal metabolism varies between individuals, but that 0.65 g per kg per day is sufficient to meet the needs of half (the median) of the population. For an average 70 kg person, this corresponds to only 45 g of protein, the equivalent of a serving of yogurt for lunch and a tuna sandwich for dinner. However, since half of the population requires a higher intake due to various factors (muscle mass, level of physical activity, age), a quantity of protein that corresponds to 2 standard deviations from the median is recommended to cover the needs of 97.5% of the population (Figure 1). This recommended dietary intake of protein (0.8 g per kg per day, i.e., about 50 g of protein for a 70 kg person) is therefore already higher than the basic needs of a large part of the population. In wealthy societies like ours, the likelihood of having a protein deficiency is therefore very low.
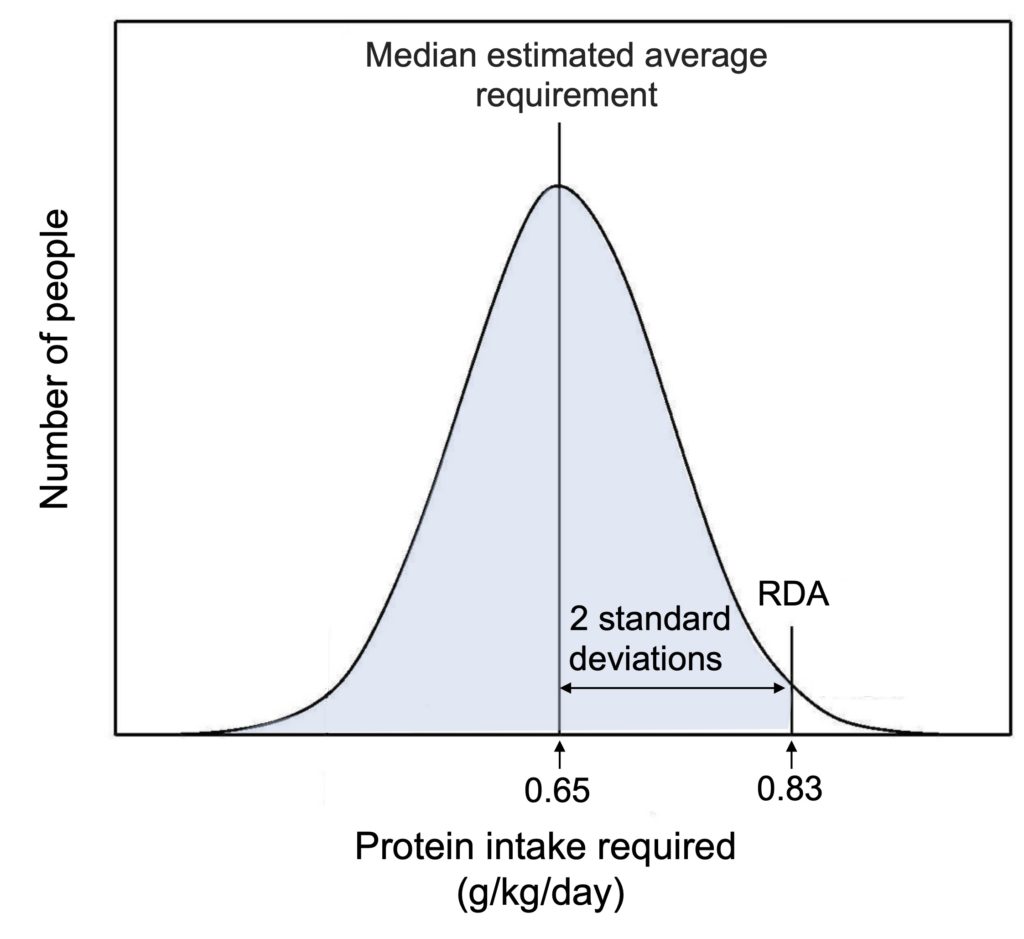
Figure 1. Inter-individual variation in the amounts of protein required to meet requirements in healthy people. The curve schematically illustrates the results of studies that measured the minimum amount of dietary protein needed to compensate for the losses associated with normal metabolism. These studies have determined the estimated average protein requirement, i.e., the amount that meets the needs of half (median) of the population (0.65 g/kg/day). To determine the recommended dietary allowance (RDA), which will cover the needs of the vast majority (97.5%) of the population (coloured area), the value corresponding to two standard deviations from the median (0.83 g/kg/day) is used.
Lots of protein
This is especially true when looking at the distribution of daily protein intake typical of the North American population (Figure 2). Only 15% of people consume less protein than the RDA (but still above the estimated average requirement), while the vast majority of people (85%) exceed this threshold, with almost a quarter of the population consuming twice as much protein as the recommended amounts.
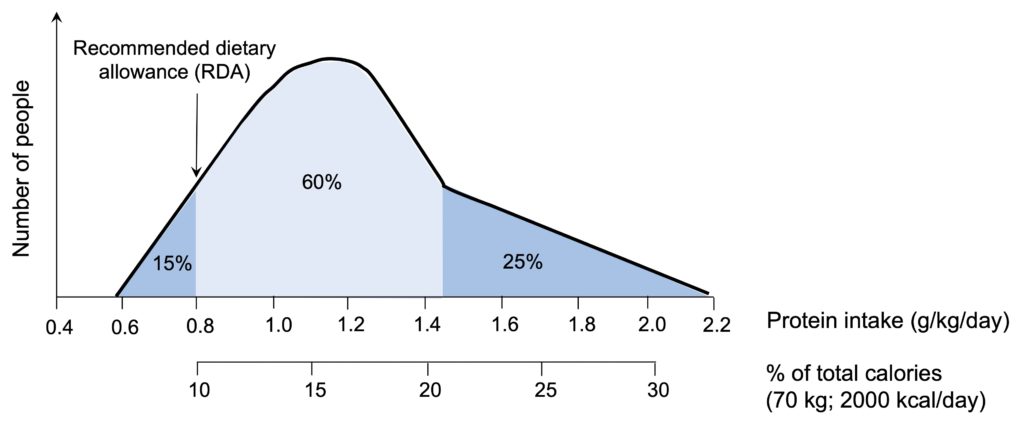
Figure 2. Distribution of daily protein intake in the US population. Note that 85% of the population consumes more than the recommended dietary allowance (RDA) of 0.8 g per kg per day, or approximately 50 g of protein for a 70 kg person. Nearly 25% of the population consumes even double the RDA, which corresponds to more than 20% of the average total caloric intake. Adapted from Zhang et al. (2024).
This high protein consumption by North Americans is clear when compared to that of people in other parts of the world (Figure 3). On average, daily protein intake is around 90 g per person, two-thirds of which comes from animal sources. In Canada, more specifically, a study established that the average quantity of protein consumed is slightly lower than the North American average (80 vs. 90 g per person), but that the proportion of animal-based protein is the same, i.e., two thirds of protein comes from red meats and deli meats (22%), poultry and eggs (20%), dairy products (17%), and fish and seafood (5%), compared to only 30% from plant sources (mainly cereals) (Figure 4).
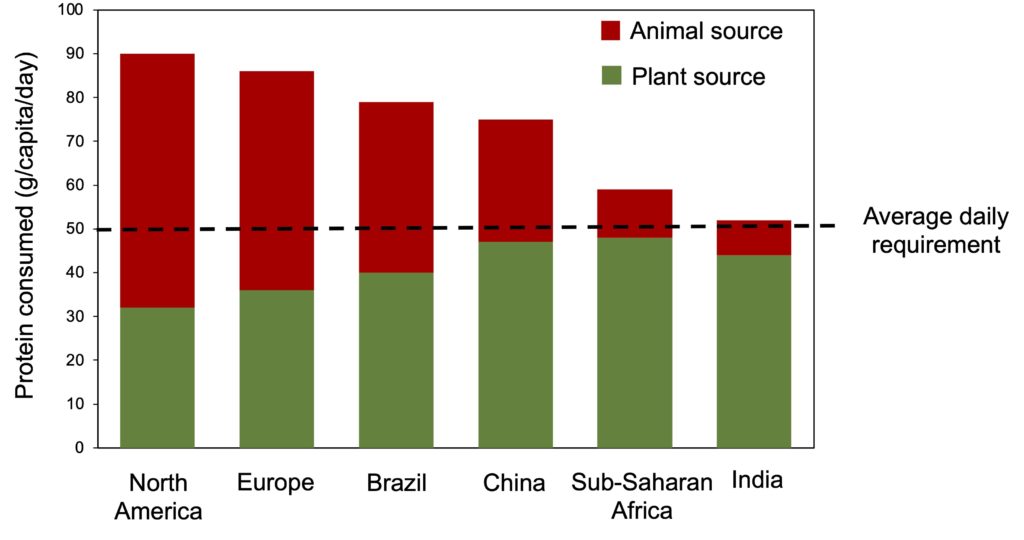
Figure 3. Comparison of average daily protein intake in different regions of the world. From World Resources Institute. Note that the required daily amount shown in the figure is the recommended dietary allowance for an average 70 kg person (0.8 g of protein per kg per day).
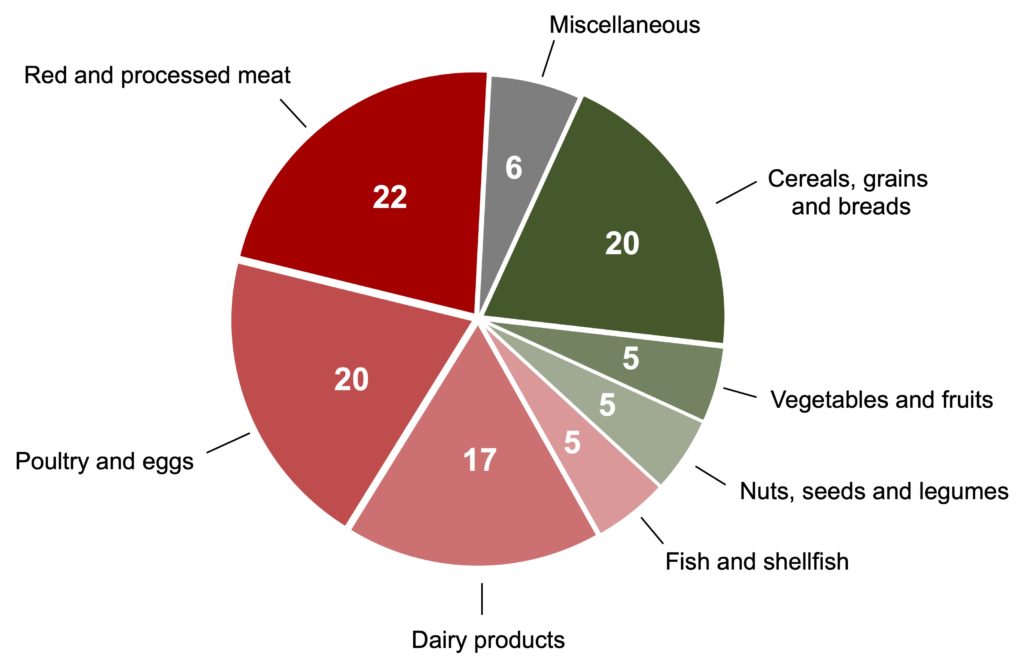
Figure 4. Distribution of dietary protein intake among Canadian adults. The values shown are percentages of total protein intake, which is around 80 g per day on average. Note that 64% of the protein intake comes from animal sources (red shades) compared to 30% from plant-based foods (green shades). From Auclair & Burgos (2021).
Overall, we can therefore see that we are the biggest consumers of protein on the planet, particularly when it comes to animal proteins. There are many problems associated with the modern Western diet (too much ultra-processed foods and red meat, and not enough plants and fibre, for example), but insufficient protein is certainly not one of them.
Protein and muscle mass
Several experts believe that higher than recommended protein intake is not harmful for healthy people and may even be necessary for very physically active people (1.2 -2 g of protein per kg recommended) or the elderly (>1.2 g per kg per day).
Maintaining or increasing muscle mass is the main reason given to justify the importance of dietary protein intake above the recommended threshold. Our metabolism can store excess sugar (in the form of glycogen) or fat (in adipose tissue) but cannot store proteins. In the case of insufficient protein intake, the missing amino acids mainly come from the muscles, which contain approximately 60% of total protein in the human body. This is particularly problematic for the elderly, who are already at risk of declining muscle mass (from the age of 50, a loss of muscle mass of the order of 0.5% to 1% per year is observed), and it is for this reason that a higher protein intake is recommended for this population.
The incentive to consume more protein is particularly pronounced among people who are training and need to recover the muscle mass lost during exercise or who seek to increase their muscularity and/or their muscular power. Protein ingestion does stimulate muscle protein synthesis, but this process quickly reaches a maximum at 0.25 g/kg in 18-37 year-olds and 0.40 g/kg in ≥ 55-year-olds. In practical terms, this means that there is a synthesis of new muscle protein up to quantities of 18 g per meal for young adults and 28 g per meal for older people, which roughly corresponds to the recommended dietary allowance for both populations. Beyond these quantities, however, excess amino acids (proteins) are oxidized, without the possibility of participating in muscle protein synthesis. Adding protein snacks between meals is not really a solution either, because muscle protein synthesis remains resistant to high blood levels of amino acids several hours after meals. In other words, even increasing the quantity and frequency of protein intake quickly leads to saturation of the system involved in muscle protein synthesis, making it difficult to increase muscle mass simply by consuming extra protein. Moreover, a large number of studies have reported that protein intake above the RDA has no measurable benefit on muscle strength or overall fitness.
Too much protein?
If quantities of protein above recommended thresholds do not seem to provide major health benefits, could they on the contrary have negative effects? For certain pathologies such as kidney disease, the answer is clearly yes: since the kidneys filter and eliminate waste from protein metabolism, a reduction in kidney function can lead to damage and an accumulation of toxic substances. Among healthy people, on the other hand, the current trend is to consider that high amounts of protein have no major impact on health. For example, the Institute of Medicine (IOM), an independent organization affiliated with the American Academy of Sciences, suggests a protein intake that can vary between 10-35% of total energy, despite the fact that intakes ≥ 15% far exceed the recommended dietary allowance (for a 70 kg person, the RDA of 50 g of protein corresponds to 200 calories, or approximately 10% of total calories consumed daily on average).
This permissive approach to high amounts of protein is somewhat surprising, given that several studies conducted in recent years have reported that excessive protein intake can have negative repercussions on health. For example, a large number of studies carried out on animal models (rabbits, rodents and great apes, among others) have clearly established a link between high protein intake and accelerated atherosclerotic plaque formation. A similar phenomenon may be at work in humans, as a Swedish study of 43,396 women followed for around fifteen years showed that a high protein intake was associated with a higher risk of cardiovascular disease.
Additionally, at least eight large population-based studies (see here and here, for representative studies) have reported that high protein consumers had an increased risk of developing type 2 diabetes compared to those who ate less protein. This negative effect is biologically plausible, as it is well established that protein stimulates insulin secretion while interfering with its signal in the muscles. Over long periods, this situation can produce insulin resistance and over time lead to pancreatic dysfunction and the onset of type 2 diabetes.
It is important to note that the increased risk of cardiovascular disease and diabetes associated with high protein intake is observed for animal proteins, but not for plant proteins. It is therefore essentially the predominance of animal proteins (two thirds of protein in Western countries) that is responsible for the negative effect of a high-protein diet on the risk of developing these pathologies.
mTOR, the conductor of the metabolic response to protein
An inherent characteristic of a diet overloaded with animal proteins is that it provides high quantities of branched-chain amino acids (BCAA : leucine, isoleucine and valine), which are recognized as powerful activators of the mTOR complex (mTORC1). When these amino acids are present in high amounts, such as when protein intake exceeds recommended thresholds, mTOR is activated and increases the synthesis of proteins, lipids and nucleotides required for growth. This activity is crucial to the production of the proteins needed to maintain muscle mass, but overactivation by mTOR can nevertheless have negative repercussions in other organs of the body and cause metabolic disturbances that can lead, for example, to insulin resistance (see our article on this subject).
At the same time, the activation of mTOR leads to a blockage of autophagy, the process by which the cell uses its own constituents as an energy source (thus avoiding a futile cycle where new cellular components manufactured in response to mTOR activation are immediately degraded). However, autophagy is essential for eliminating dysfunctional elements that accumulate over time in cells, both in terms of proteins and larger constituents such as mitochondria (this is known as mitophagy). Inhibition of autophagy by excess protein can therefore cause an accumulation of defective structures that interfere with the functioning of the cell and eventually cause its death.
It is therefore possible that an excessive intake of animal proteins, which far exceeds the recommended thresholds, can have negative effects on certain physiological processes (cardiovascular disease for example) due to this overactivation of mTOR.
Proteins and atherosclerosis: Macrophages as intermediaries
This possibility is supported by the results of a recent study that shows that high amounts of protein stimulate the mTOR complex present in a class of immune cells known as macrophages, and that this phenomenon correlates with an acceleration in the development of atherosclerosis.
This contribution of the immune system to the atherosclerosis process may seem surprising at first glance, given that it is the progressive accumulation of fat deposits, notably cholesterol carried by low-density lipoproteins (LDL), which is largely responsible for the formation of these plaques. In fact, reducing blood levels of LDL cholesterol, either through lifestyle changes (diet, exercise) or medication (statins), remains one of the main interventions used to slow down the development of atherosclerosis and thus reduce the risk of its complications, in particular myocardial infarction and stroke.
In reality, however, atherosclerosis is a much more complex pathology that does not depend exclusively on blood levels of LDL cholesterol. Clinically, for example, it is well established that even when these LDL levels are reduced to very low levels with statins, a significant percentage of patients (almost a quarter in some studies) remain at very high risk of relapse in the years following the start of treatment. We now know that this residual risk, independent of LDL cholesterol, is largely due to chronic inflammation, caused in particular by the infiltration of macrophages in the arterial wall (Figure 5).
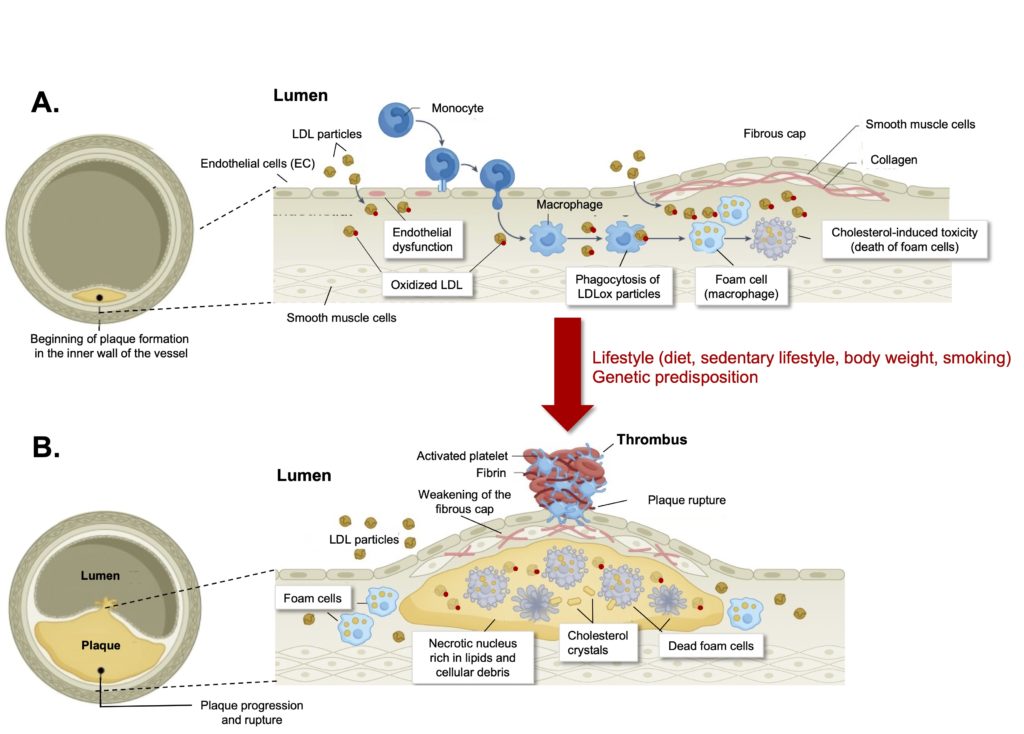
Figure 5. Central role of macrophages in the progression of atherosclerosis.
A. The attack on the layer of endothelial cells that line the internal wall of the vessel allows the entry and oxidation of cholesterol-rich LDL particles inside the wall. The pro-inflammatory effect of this influx of oxidized lipids attracts monocytes from the circulation through the expression of certain surface receptors (VCAM/ICAM-1), which allows them to infiltrate the wall. The monocytes then transform into macrophages and ingest nearby oxidized LDL particles by phagocytosis. When the quantities of LDL taken up by macrophages are large, they become overloaded with lipids and transform into foam cells. This overload is toxic to the cells and leads to their death and the release of the lipids they contain. This phenomenon leads to a chronic local inflammatory reaction, which stimulates the growth of muscle cells in the vessel wall, resulting in the formation of a fibrous cap that isolates the dead or dying macrophages and fatty material from the bloodstream. These early plaques do not present any immediate danger, as the fibrous cap is very stable and is able to hermetically seal the fatty deposits present in the artery wall.
B. The continuous attack on endothelial cells, whether due to chronic exposure to high quantities of LDL particles or to certain lifestyle factors that promote inflammation (obesity, sedentary lifestyle, smoking), leads over time to an accumulation of foam cells and the progression of atherosclerosis. Typically, a necrotic core is observed consisting of dead cells and lipids, including cholesterol crystals. The highly pro-inflammatory climate generated within this lipid core can then destabilize the fibrous layer, causing the appearance of very thin, fragile areas on the surface of the plaque. The rupture of these plaques then exposes their contents to the coagulation system, which, believing that it is an injury to be repaired, immediately forms a thrombus (clot) that completely obstructs the vessel, leading to infarction (coronary blockage) or stroke (carotid blockage). Adapted from Chen et al. (2022).
It all begins when the endothelial cells lining the artery walls are confronted with an imbalance that affects their function (known as endothelial dysfunction), whether due to an excess of LDL particles or to various inflammatory stimuli (smoking, microorganisms, chronic inflammatory conditions linked to lifestyle such as being overweight and sedentary). This protective layer then loses its tightness, which will facilitate the entry of LDL particles and their subsequent oxidation by free radicals produced by dysfunctional endothelial cells. At the same time, the expression of receptors on the surface of the endothelium enables a class of immune cells, the monocytes, to infiltrate the vessel wall, where they transform into macrophages. These macrophages play a key role in the progression of atherosclerosis by capturing large quantities of oxidized LDL by phagocytosis, which transforms them into foam cells in which cholesterol deposits can be observed (the term “foam”, from the Latin “spuma”, derives from the large quantity of lipids they contain, which appear as “bubbles” when the cells are examined by microscopy).
Chronic exposure of the vessel wall to conditions that promote the invasion of the arterial wall by macrophages (lifestyle, certain genetic predispositions) creates, over time, an accumulation of foam cells and the formation of a lipid core in the centre of the plaque, composed of cholesterol and cellular debris. The highly inflammatory climate within this necrotic area can gradually destabilize the fibrous cap, making it more fragile and at risk of cracking. When this rupture occurs, the leakage of the plaque contents is perceived by the coagulation system as a wound to be sealed, leading to the aggregation of platelets, the formation of a fibrin network, and the appearance of a clot that will obstruct the passage of blood through the artery.
In sum, the progression of atherosclerosis is not strictly a consequence of high cholesterol levels: chronic inflammation caused by the accumulation of lipid-saturated macrophages also plays a key role in this process. The activation of these macrophages could therefore represent a biologically plausible mechanism to explain the increased risk of cardiovascular disease associated with high protein intake.
Activating proteins
To determine the amount of dietary protein associated with macrophage activation, researchers gave volunteers two different meals, containing either 15% or 22% of total calories in the form of protein. These quantities were chosen because one of them (15%) corresponds to the protein intake of most people, slightly above the recommended minimums, while the other (22%) corresponds to double the recommended intake and is representative of the high protein consumption by a significant proportion of the population (see Figure 2).
The activity of the mTOR complex in monocytes isolated from the participants’ blood was subsequently evaluated by measuring two reactions that are the signature of an activated mTOR complex, namely the transfer of a phosphate group to an enzyme involved in the synthesis of new proteins and cell growth (S6 protein), as well as to an enzyme that blocks the autophagy process (ULK1) (Figure 6). The results clearly show that a high protein intake, much higher than that recommended, has a stronger influence on mTOR activity, both by increasing its activating effect on growth (phosphorylation of S6) and by reducing the cell autophagy process.
Intriguingly, although several amino acids can activate mTOR, the researchers observed that leucine alone was able to replicate the effect of proteins on the activation of cell growth pathways and the inhibition of autophagy. (Figure 6 C).
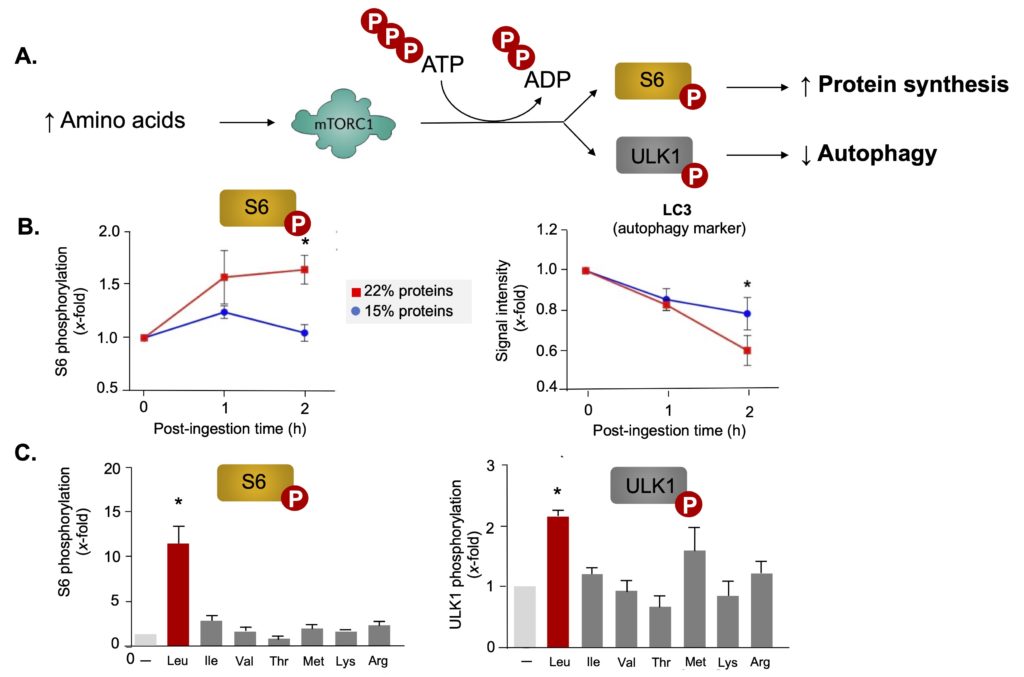
Figure 6. Involvement of the mTOR complex in the metabolic response to a high-protein diet.
A. Schematic representation of methods used to measure mTORC1 activity in cells. In response to the presence of large quantities of amino acids from protein digestion, mTOR is activated and transfers a phosphate group from ATP to certain proteins in the cell. Phosphorylation of the S6 protein participates in the cascade leading to new protein production and growth, while phosphorylation of the ULK1 protein contributes to the blocking of the autophagy process.
B. High dietary protein intake activates the mTOR complex and inhibits autophagy in human monocytes. Nine fasted volunteers consumed meals containing 15% or 22% of total calories as protein. Additional protein came from a combination of eggs, chicken, beef, and whey. Monocytes from the volunteers’ blood were isolated just before the intervention or 1 and 2 hours after ingestion of the meals, and the cells’ mTOR activity was measured by the increase in phosphorylation of the S6 protein (left graph) or by analysis of the distribution within cells of the LC3 protein, a marker of the autophagic process (right graph).
C. Leucine is the main amino acid responsible for the activation of mTOR induced by high protein intake. Cultured human monocytes were exposed to 7 amino acids whose blood concentrations are most strongly increased after ingestion of a protein-rich meal. mTOR activity was assessed by measuring the phosphorylation of the S6 protein (left graph) or the ULK1 protein (right graph). Asterisks indicate a statistically significant difference. From Zhang et al. (2024).
Atherosclerosis is a gradual process, the development of which can take place over several decades, and it is therefore impossible to quantify in a short-term study the impact of a high-protein diet on the evolution of atherosclerosis plaques in humans. To overcome this limitation, the researchers used mouse models fed diets containing proportions of protein equivalent to those administered to the study volunteers (15% and 22% of total calories), which similarly stimulate the mTOR activity in monocytes/macrophages.
After only 8 weeks of a high-protein diet, they observed that the number of plaques in the aorta was greatly increased, and that these were infiltrated by a greater number of macrophages. The volume of the necrotic area in the centre of the plaques, containing cellular debris, was also much larger, most likely due to a large number of dead foam cells. According to previous results obtained by the same group, it is very likely that this cell death is a direct consequence of the inhibition of autophagy associated with the activation of mTOR, more precisely mitophagy (mitochondrial autophagy).
All these results suggest that high protein intake leads to overactivation of mTOR, death of macrophages infiltrated in the arterial wall, and accelerated development of atherosclerosis. However, the amount of protein required to start this process is quite high, well beyond the recommended dietary allowance of 0.8 g per kg per day (Figure 7). Higher daily intakes, such as those recommended for active and/or elderly people (1-1.2 g per kg) also do not seem to have any major effect on cardiovascular risk, which is reassuring. On the other hand, data suggest that the 20-25% of the population who consume very high amounts of protein (≥ 1.4 g per kg, or approximately ≥ 20% of total calories) have a much higher cardiovascular risk.
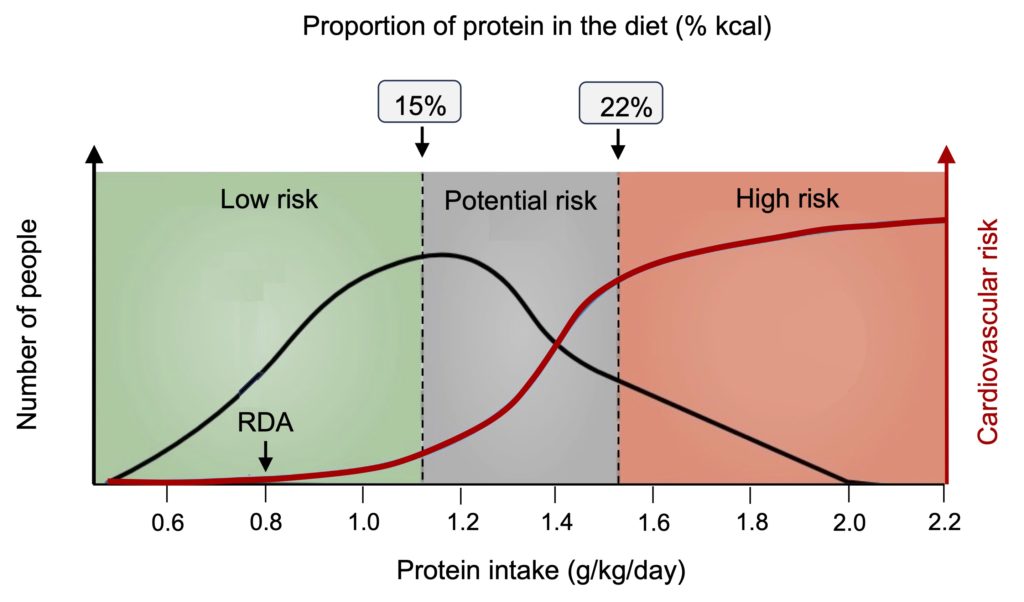
Figure 7. Estimation of cardiovascular risk based on daily protein intake. The results of the study indicate that a significant increase in the risk of atherosclerosis and cardiovascular disease (red line) requires high quantities of protein, beyond 1.4-1.5 g per kg per day, i.e., almost twice the recommended dietary allowance (RDA). According to the distribution curve of protein intake in the population (black line), this risk nevertheless affects almost a quarter of the population. Adapted from Zhang et al. (2024).
More plant-based protein
A very important point raised by the results of the study is that the atherogenic effect of high amounts of protein appears to be primarily caused by a single amino acid, leucine. This is particularly interesting because leucine is much more abundant in animal proteins than in plant-based proteins (see Table 1), which could explain why only animal proteins have been associated with an increased risk of cardiovascular disease and type 2 diabetes in epidemiological studies. In addition to respecting the recommended dietary allowance (between 0.8 and 1.2 g per kg per day), an obvious way to reduce the negative impact of high protein intake is to better distribute protein sources, by increasing the proportion that comes from plant-based foods. It’s well worth the effort: one study, for example, showed that replacing just 3% of calories from animal proteins (red meats and processed meats, in particular) with the equivalent in plant-based proteins is associated with a significant reduction (34%) in the risk of premature mortality.
Table 1. Leucine content of different commonly consumed foods. Data are from Rondanelli et al. (2021) and the USDA Nutrient Database-Leucine (2018). Note that the recommended dietary allowance for leucine is 42 mg per kg per day, or approximately 3000 mg for an average 70 kg adult. The increase in cardiovascular risk reported in the study is observed at 1.5 g of protein per kg (100 g for a 70 kg adult), which corresponds to approximately 9000 mg of leucine, three times the recommended intake.
| Animal sources | Plant sources | ||||
|---|---|---|---|---|---|
| Food | Portion | Leucine (mg) | Food | Portion | Leucine (mg) |
| Beef (sirloin) | 85 g | 2180 | Tofu | 100 g | 1370 |
| Chicken (thigh) | 85 g | 2031 | Pasta | 100 g | 1033 |
| Tuna (can) | 85 g | 1883 | Bread | 100 g | 691 |
| Chicken (breast) | 85 g | 1661 | Peanut butter | 30 g | 440 |
| Cooked ham | 85 g | 1440 | Green peas | ½ cup | 234 |
| Salmon | 85 g | 1272 | Corn | ½ cup | 207 |
| Cheese (cheddar) | 50 g | 1000 | Rice (long-grain) | ½ cup | 183 |
| Greek yogurt | ¾ cup | 707 | Orange juice | 1 cup | 139 |
| Egg | 1 | 541 | Broccoli | ¾ cup | 133 |
| Milk (2%) | 1 cup | 377 | Tomatoes | ½ cup | 45 |
Unfortunately, there is still a lot of reluctance to adopt plants as a source of protein. There is a widespread misconception that amino acid intake may be insufficient in vegetarian diets, yet there is clear evidence that the quantities and proportions of amino acids consumed by vegetarians and vegans are generally more than sufficient to meet and exceed individual daily requirements, provided that a reasonable variety of foods is consumed and that energy needs are met. The belief that some plant foods “lack” specific amino acids is false: all plant foods contain all 20 amino acids, including all 9 essential amino acids. It is the distribution profile of some of these amino acids that is less optimal in plant foods than in those of animal origin. For example, lysine is present in cereals in proportions well below the optimal proportions for human needs, while the amounts of sulfur-containing amino acids (methionine and cysteine) in legumes are slightly below the optimal intake. To be deficient in certain amino acids, a person would therefore have to eat only rice or beans every day, which is obviously unlikely. In reality, a normal diet, even if minimally varied (as is generally the case in wealthy countries), provides an adequate supply of all essential amino acids. It is also worth noting that the inhabitants of the island of Okinawa, home to the highest proportion of centenarians in the world, eat relatively little protein (9% of total calories), and that a high proportion of this protein comes from plants.
