Dr Louis Bherer, Ph. D., Neuropsychologue
Professeur titulaire, Département de Médecine, Université de Montréal, Directeur adjoint scientifique à la direction de la prévention, chercheur et Directeur du Centre ÉPIC, Institut de cardiologie de Montréal.
See all articlesDr Martin Juneau, M.D., FRCP
Cardiologue, directeur de l'Observatoire de la prévention de l'Institut de Cardiologie de Montréal. Professeur titulaire de clinique, Faculté de médecine de l'Université de Montréal. / Cardiologist and Director of Prevention Watch, Montreal Heart Institute. Clinical Professor, Faculty of Medicine, University of Montreal.
See all articlesOverview
- Exerkines are molecules released in response to acute and/or chronic exercise, which have effects on various signalling pathways, endocrine, paracrine and/or autocrine.
- Different types of organs, cells and tissues release exerkines, including skeletal muscle (myokines), heart (cardiokines), liver (hepatokines), white adipose tissue (adipokines), brown adipose tissue (batokines), and neurons (neurokines).
- Exerkines are present in several forms: hormones, metabolites, proteins, and nucleic acids.
- The multitude of exerkines released during exercise and the wide variety of organs that release them or are influenced by these molecules would explain why the response to exercise is so variable and complex.
It is well established that physical activity, exercise and cardiorespiratory fitness are very important for preventing and treating chronic diseases such as cardiovascular disease, obesity, type 2 diabetes, cognitive decline, and several cancers. In addition, regular exercise improves the immune system and increases life expectancy, particularly healthy life expectancy (Figure 1). Conversely, physical inactivity is associated with the development of several chronic diseases, a reduction in life expectancy, and a significant economic burden on society.
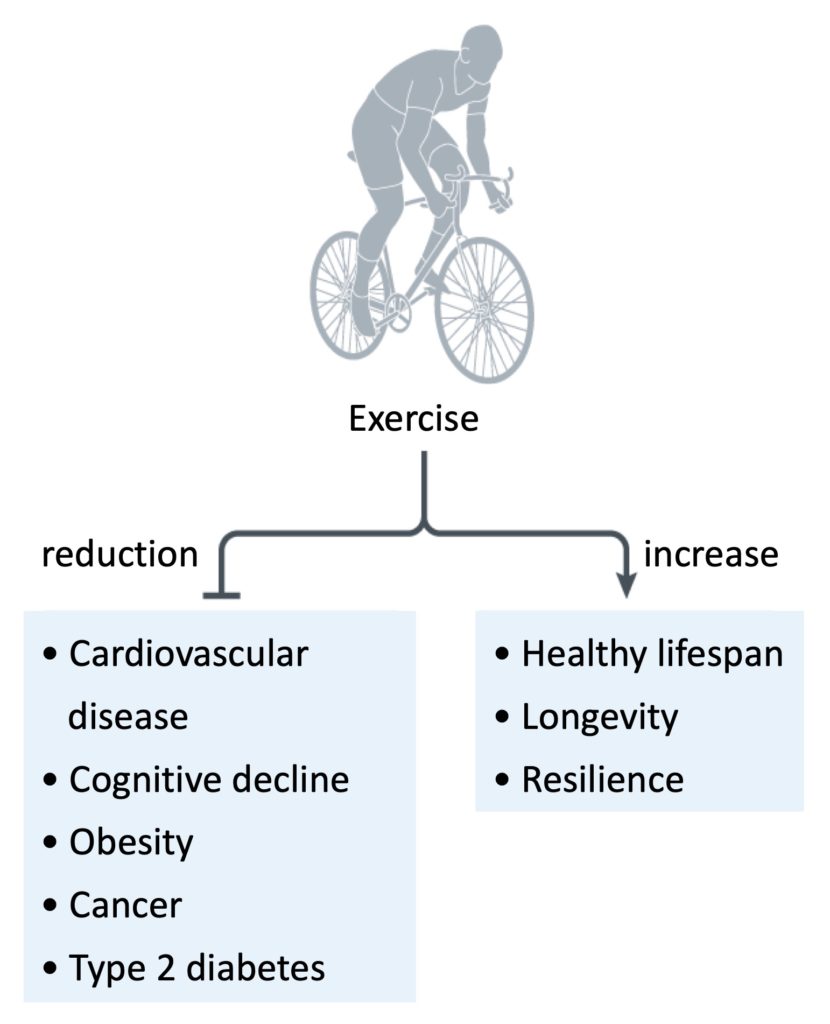
Figure 1. Exercise has many health benefits, including reductions in the incidence and severity of certain diseases, in addition to increased longevity and resilience. Adapted from Chow et al., 2022.
Exerkines
Exerkines are molecules released in response to acute and/or chronic exercise and that have effects on various signalling pathways (see this review article). The term “exerkine” was coined in 2016, but it has long been known that factors are released during exercise, with lactic acid produced by skeletal muscles being the best-known example. Subsequently, the beneficial effects of exercise were attributed to hormones released by muscles: myokines. Since skeletal muscles represent approximately 1/3 of body weight and play a crucial role in exercise, research initially focused on myokines, particularly interleukin-6 (IL- 6) following its discovery in 2000. Subsequently, exerkines released by other organs during exercise were identified: cardiokines (heart), hepatokines (liver), adipokines (white adipose tissue), batokines (brown adipose tissue), and neurokines (neurons).
Variability of response to exercise
The physiological response to exercise is highly variable in humans and animals and is influenced by several external and internal factors. Among the external factors, there is the circadian rhythm, whether or not you are fasting, or even the composition of the post-exercise meal. Among internal factors, genetics play an important role in the response to exercise. According to one study, around 20% of people do not improve their respiratory capacity when they exercise regularly; they are referred to as “non-respondents”. Additionally, 7 to 15% of people experience adverse reactions when exercising on a regular basis: increased systolic blood pressure, increased blood levels of triglycerides and insulin, and lower HDL cholesterol (“good cholesterol”). The role of exerkines in the variability of the response to exercise is the subject of ongoing studies.
The release of exerkines in response to acute exercise is influenced by the type of exercise, the duration of the exercise session, physical fitness, fasting or not, and the timing of the blood sample collection after exercise. Exerkines typically released into the blood during acute exercise include interleukins 6, 8, 10 (IL-6, IL-8, IL-10) and IL-1 receptor antagonist (IL-1RA). One study showed that blood levels of several cytokines (IL-6, IL-1RA, IL-10, and tumour necrosis factor (TNF)) are higher after a marathon, reaching a peak value 1 to 2 hours after exercise and remain elevated approximately 4 hours. The higher the intensity of exercise, the higher the blood levels of exerkines after exercise.
The release of exerkines in response to a single exercise session (acute exercise) is not identical to that observed in response to chronic exercise. Acute exercise is usually associated with responses focused on maintaining metabolic homeostasis (balance), where the effect of pro-inflammatory exerkines is counterbalanced by the concomitant release of anti-inflammatory factors and where sources of energy (glycogen, glucose, fat) change according to needs. Chronic exercise, however, is associated with responses focused on long-term metabolic adaptations and reduced inflammation (reduced concentration of IL-6 in the blood, for example).
Autocrine, paracrine and endocrine effects
Research on exerkines initially looked at changes in the levels of cytokines (IL-6 in particular) in the blood, before and after an exercise session. Researchers then examined the effects of exerkines released by skeletal muscle on other organs (endocrine effect). More recent research has focused on the effects of exerkines on the very tissue that secreted it (autocrine effect), on tissues located in the immediate environment (paracrine effect), on exerkines released by organs other than muscles, and on the profiling of all exerkines rather than on a particular exerkine.
An example of an autocrine effect is the release of lactate, musclin, and myostatin by the myocytes of skeletal muscle, which stimulates the biogenesis of mitochondria in this tissue (to be able to produce more energy) and modulates the use of energy sources. The exerkines VEGF, angiopoietin 1 and IL-8, released by the same skeletal muscle during exercise, have effects in the immediate environment (paracrine effect): induction of angiogenesis (formation of new blood vessels), modulation of blood flow, and increase in nutrient availability to support muscle tissue growth. Other paracrine effects have been observed in the nervous system, adipose tissue, bone, cartilage, extracellular matrix, and the immune system.
Exerkines and the cardiovascular system
Animal and human studies indicate that exerkines can improve cardiometabolic health. The contraction of skeletal muscles releases several molecules favourable to the cardiovascular system (Figure below). The exerkines angiopoietin 1 (see here, here and here), fractalkine (see here and here), fibroblast growth factor 21 (FGF21) (see here and here), IL-6, IL-8 (see here and here), musclin, myonectin, and vascular endothelial growth factor (VEGF) (see here, here and here) are generally released during exercise.
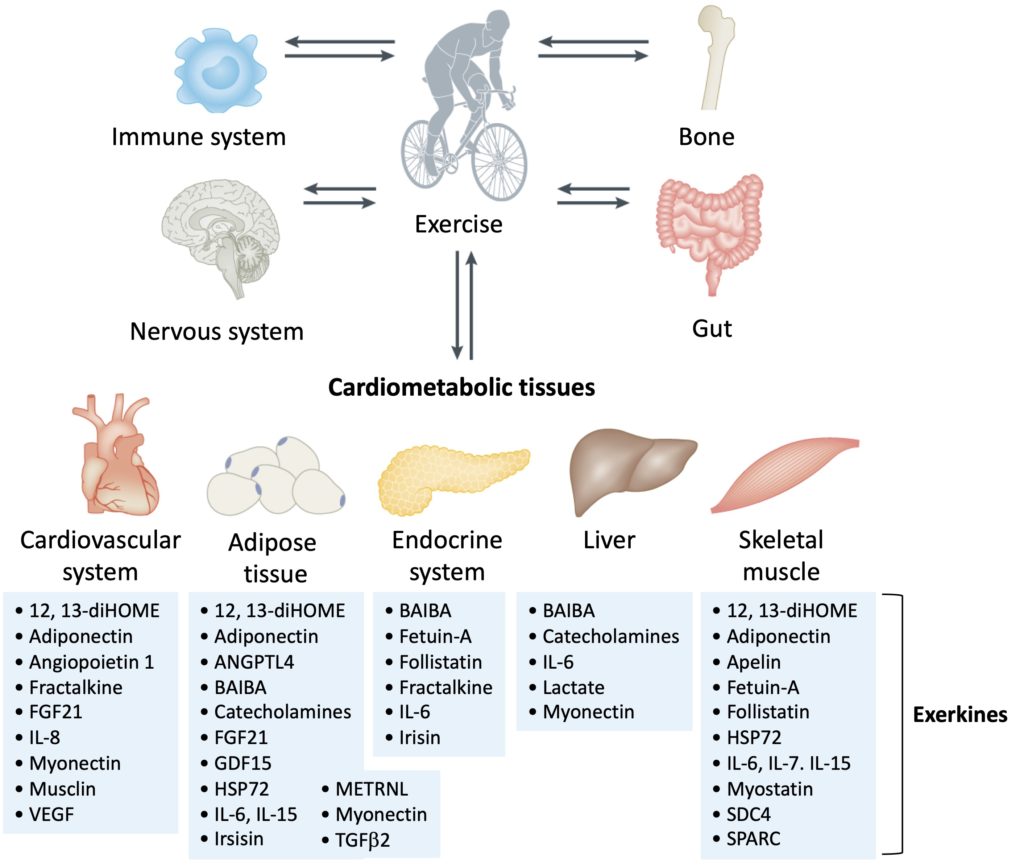
Figure 2. Examples of exerkines that act on the cardiometabolic system. Exerkines are released into the bloodstream by several organs and tissues during and after exercise. These molecules have varied effects on five cardiometabolic tissues, which are sources of exerkines and are directly affected by exercise. Adapted from Chow et al., 2022.
In the cardiovascular system, exerkines promote vascularization and angiogenesis (formation of new blood vessels), in addition to improving blood pressure, endothelial function and overall fitness. In adipose tissue, exerkines increase the uptake of fatty acids, improve lipolysis (degradation of lipids into fatty acids), thermogenesis (heat production), and glucose metabolism. In the liver, exerkines improve glucose metabolism and fatty acid uptake. In skeletal muscles, exerkines promote muscle growth, maintenance and repair of muscle fibres, glucose uptake, lipid oxidation, mitochondrial biogenesis, and the formation of new blood capillaries in the muscles. In the pancreas, exerkines improve cell viability and influence insulin secretion.
Adipose tissue
Exercise stimulates lipolysis in white adipose tissue (white fat), releasing free fatty acids that can be used as an energy source. This effect has mainly been attributed to the release of adrenaline, but also, to a lesser extent, to the release of growth and differentiation factor 15 (GDF15) and IL-6 during acute exercise. Another potential effect of exercise on white adipose tissue is “browning” (transformation of tissue with the characteristics of brown adipose tissue), by increasing mitochondria numbers, metabolic rates, and heat production. Studies in mice suggest that the release of irisin caused the “browning” of white adipose tissue and a reduction in obesity, but studies in humans do not support such a conclusion. For example, one study did not find “browning” of adipose tissue (biopsy analysis) in people subjected to chronic physical training.
Adipose tissue can also secrete exerkines, such as 12,13-dihydroxy-9Z-ocatadecanoic acid (12,13-diHOME), which is released by brown adipose tissue and increases the oxidative capacity of skeletal muscle. The results of some studies suggest that a sustained high level of 12,13-diHOME in the blood preserves cardiac function and remodeling and increases cardiac hemodynamics through a direct effect on cardiomyocytes. Additionally, reduced levels of 12,13-diHOME in the bloodstream have been associated with an increased incidence of cardiovascular disease.
The release of lactate from skeletal muscle also impacts the response of adipose tissue to exercise. For example, in mice, exposure to lactate stimulates the secretion of transforming growth factor 2 (TGFb2) by adipocytes. In the same study, when mice exercised regularly, the level of TGFb2 increased significantly in adipocytes, and this increase was associated with improvements in glucose metabolism, lipid oxidation, and a possible reduction of inflammation of adipose tissue. Similar results were found in people who exercised regularly, although to a lesser degree than in animal models.
Skeletal muscle
Exerkines released into the bloodstream by multiple tissues can improve skeletal muscle function and growth. For example, in an animal model, apelin improves muscle function during aging, suggesting that this exerkine could potentially be used as a therapy for age-related sarcopenia (see here, here and here). Daily injections of apelin into aged mice stimulated mitochondrial biogenesis and protein synthesis in muscle cells, in addition to improving muscle regeneration stimulated by muscle stem cells.
12,13-diHOME, a batokine released during exercise by brown adipose tissue, stimulates the uptake and oxidation of free fatty acids by skeletal muscle. The hepatokines follistatin and fetuin-A released by the liver during exercise also have effects on skeletal muscle, such as stimulating muscle tissue growth and improving blood sugar control (see here, here, here and here). Other exerkines stimulate the growth and development of muscle tissue, such as IL-7, IL-15, leukemia inhibitory factor, syndecan-4, and myostatin.
Liver
Exercise reduces fatty liver disease (harmful fat buildup in the liver) regardless of weight loss. The liver is an organ that secretes thousands of proteins into the bloodstream, including several exerkines that have beneficial effects on glucose and lipid metabolism, on the “browning” of white adipose tissue in mice by growth factor fibroblast 21 (FGF21), on lipolysis in humans and mice by FGF21, and the maintenance of cellular homeostasis by the heat shock protein HSP72.
Immune system
Exercise causes an increase in lipid oxidation, mitochondrial biogenesis and local injury, which stimulates the release of exerkines into the bloodstream that act on the immune system. An exercise session increases the levels of cytokines such as TGFb1 and IL-6. This increase generates acute inflammation, characterized by an increase in tumour necrosis factor (TNF) and IL-6. After the acute phase of exercise, anti-inflammatory cytokines (such as IL-10 and IL-1RA) are released in response to acute inflammation. Regular (chronic) exercise is associated with a reduction in systemic and tissue inflammation, as well as reduced levels of TNF and IL-6 at rest, compared to sedentary people. Reductions in insulin resistance and tumour growth have been attributed to the anti-inflammatory effects of regular exercise.
The effects of exerkines on the immune system may have protective effects against cancer. A meta-analysis of 12 prospective studies found that physical activity is associated with a lower incidence of several types of cancer, and several associations remained valid even after adjusting the data for body mass index. In an animal model where tumours were implanted in mice, those that exercised had 60% less tumour growth, compared to sedentary mice. Adrenaline and IL-6 released during exercise induced the mobilization of Natural Killer (NK) cells, with tumour redistribution and infiltration slowing tumour growth.
The nervous system
Exercise helps maintain and improve brain function. The effects of exercise on the brain are particularly apparent in the hippocampus, which is involved in learning and memory. A growing number of studies indicate that exercise can prevent or slow cognitive decline. In studies that transferred blood plasma from exercised animals to sedentary animals, improvements in cognitive function were found, suggesting that a factor released during exercise can reach the brain and exert beneficial effects (see here and here). In mice, adiponectin secreted by adipocytes crosses the blood-brain barrier and is associated with increased neurogenesis and a reduction in behaviours associated with depression. The mechanisms underlying the beneficial effects of exercise on the brain, still little understood, are currently the subject of much research.
Bones
Exercise, especially resistance exercise, increases bone density. There are several mechanisms involved in this phenomenon, but mechanical load is considered to be the major factor. Certain myokines have been shown to have effects on bones: apelin, myostatin, irisin, IL-6, IL-7, and beta-aminoisobutyric acid (BAIBA).
Limitations of exerkine research
There is some controversy about exerkines, and excess enthusiasm about the effects of these compounds should be tempered. Indeed, studies on exerkines show inconsistencies between the results obtained with acute exercise and chronic exercise, between those obtained in animals and in humans, and there is great variability in the methodology of the studies, which hinders the interpretation of all the results.
Conclusions
The benefits of exercise on several systems and organs are numerous, but the underlying mechanisms remain poorly understood, particularly with regard to the great variability of the response to exercise. Although research on exerkines was initially focused on skeletal muscle, current research is much broader and includes various sources and targets of exerkines that contribute to maintaining good health. Much work remains to be accomplished and the methodology of future studies will need to be refined so that the results of the research can be translated into practical applications. Ongoing large-scale studies should provide a better understanding of the complex effects of exerkines on different systems and organs. Exerkines represent a very promising area of exercise research, which could lead to the identification of new biomarkers and will facilitate the development of exercise programs to improve health and prevent certain diseases.
