Dr Martin Juneau, M.D., FRCP
Cardiologue, directeur de l'Observatoire de la prévention de l'Institut de Cardiologie de Montréal. Professeur titulaire de clinique, Faculté de médecine de l'Université de Montréal. / Cardiologist and Director of Prevention Watch, Montreal Heart Institute. Clinical Professor, Faculty of Medicine, University of Montreal.
See all articlesOverview
- Metabolomic screening has identified a new metabolite associated with cardiovascular disease in the blood of people with type 2 diabetes.
- This metabolite, phenylacetylglutamine (PAGln), is produced by the intestinal microbiota and the liver, from the amino acid phenylalanine from dietary proteins.
- PAGln binds to adrenergic receptors expressed on the surface of blood platelets, which results in making them hyper-responsive.
- A beta blocker drug widely used in clinical practice (Carvedilol) blocks the prothrombotic effect of PAGln.
A research group from the Cleveland Clinic in the United States recently identified a new metabolite of the microbiota that is clinically and mechanistically linked to cardiovascular disease (CVD). This discovery was made possible by the use of a metabolomic approach (i.e. the study of metabolites in a given organism or tissue), a powerful and unbiased method that identified, among other things, trimethylamine oxide (TMAO) as a metabolite promoting atherosclerosis and branched-chain amino acids (BCAAs) as markers of obesity.
The new metabolomic screening has identified several compounds associated with one or more of these criteria in the blood of people with type 2 diabetes: 1) association with major adverse cardiovascular events (MACE: myocardial infarction, stroke or death) in the past 3 years; 2) heightened levels of type 2 diabetes; 3) poor correlation with indices of glycemic control. Of these compounds, five were already known: two which are derived from the intestinal microbiota (TMAO and trimethyllysine) and three others that are diacylglycerophospholipids. Among the unknown compounds, the one that was most strongly associated with MACE was identified by mass spectrometry as phenylacetylglutamine (PAGln).
In summary, here is how PAGln is generated (see the left side of Figure 1):
- The amino acid phenylalanine from dietary proteins (animal and plant origin) is mostly absorbed in the small intestine, but a portion that is not absorbed ends up in the large intestine.
- In the large intestine, phenylalanine is first transformed into phenylpyruvic acid by the intestinal microbiota, then into phenylacetic acid by certain bacteria, particularly those expressing the porA
- Phenylacetic acid is absorbed and transported to the liver via the portal vein where it is rapidly metabolized into phenylacetylglutamine or PAGln.
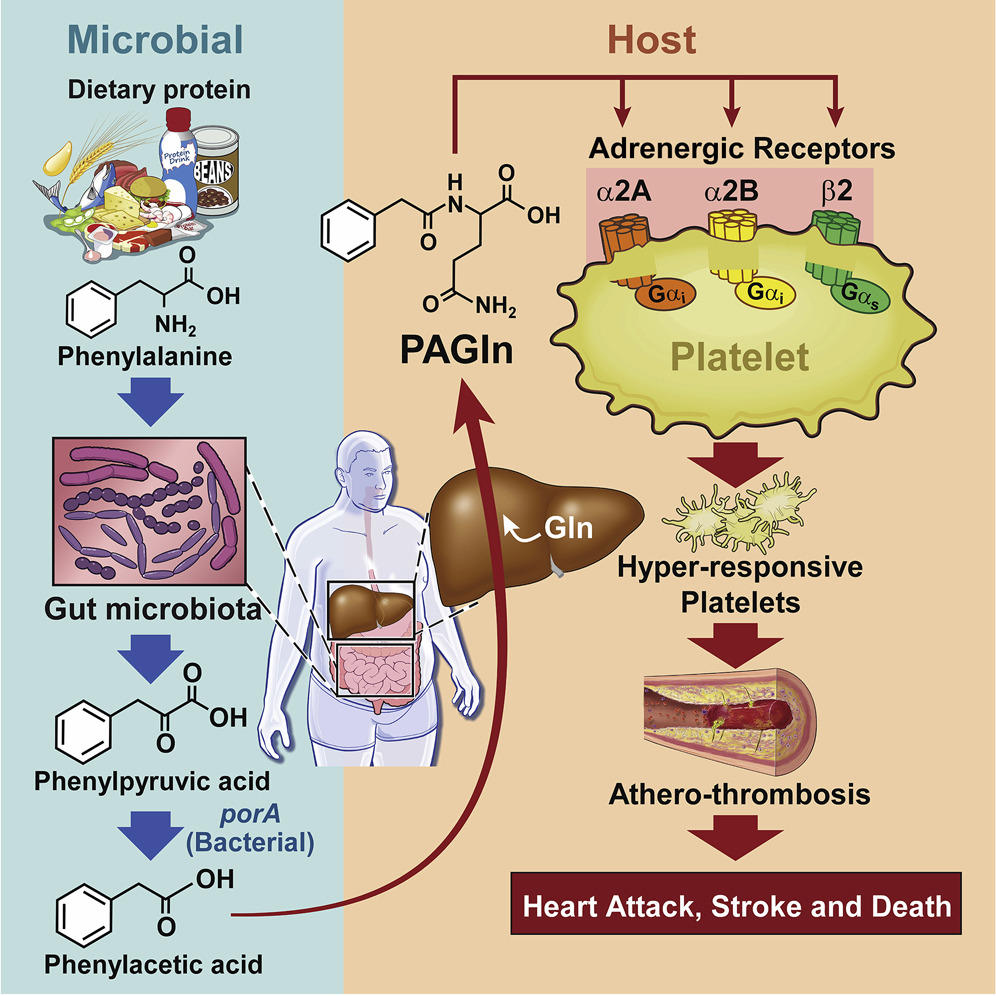
Figure 1. Schematic summary of the involvement of PAGln in the increase in platelet aggregation, athero-thrombosis and major adverse cardiovascular events. From Nemet et al., 2020.
Researchers have shown that PAGln increases the effects associated with platelet activation and the potential for thrombosis in whole blood, on isolated platelets and in animal models of arterial damage.
PAGln binds to cell sites in a saturable manner, suggesting specific binding to membrane receptors. The researchers then demonstrated that PAGln binds to G-protein coupled adrenergic receptors, expressed on the surface of the platelet cell membrane. The stimulation of these receptors by PAGln causes the hyperstimulation of the platelets, which then become hyper-responsive and accelerate the platelet aggregation and the thrombosis process.
Finally, in a mouse thrombus model, it has been shown that a beta blocker drug widely used in clinical practice (Carvedilol) blocks the prothrombotic effect of PAGln. This result is particularly interesting because it suggests that the beneficial effects of beta blockers may be partly caused by reversing the effects of high PAGln levels. The identification of PAGln could lead to the development of new targeted and personalized strategies for the treatment of cardiovascular diseases.
